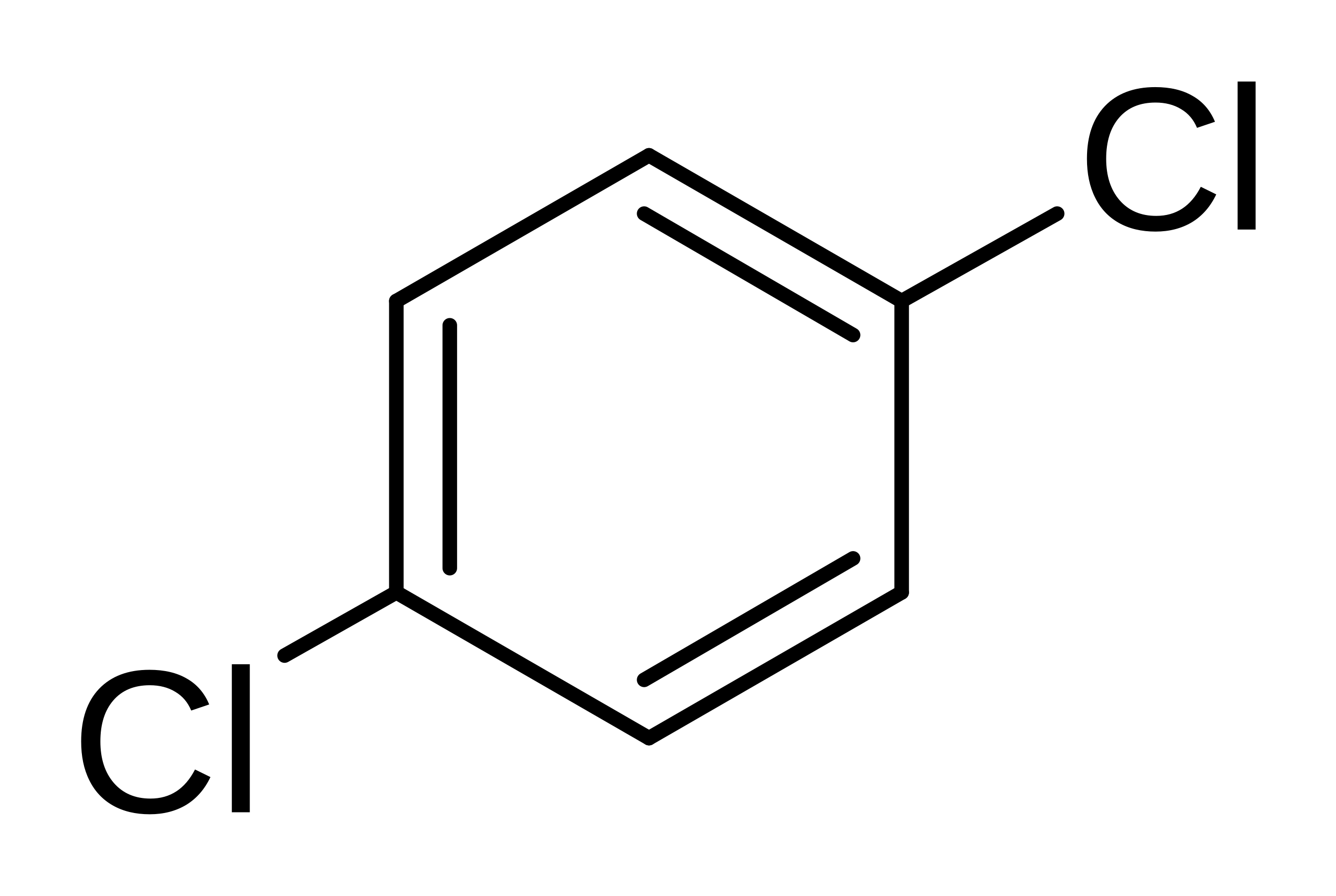
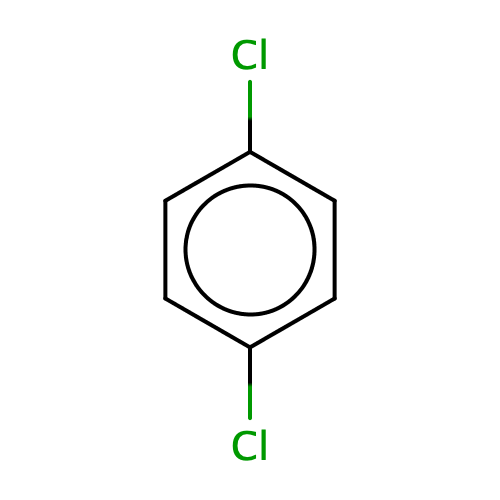
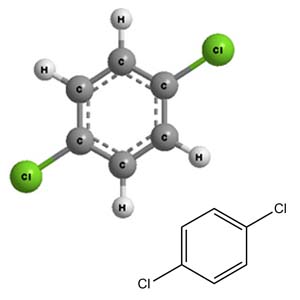
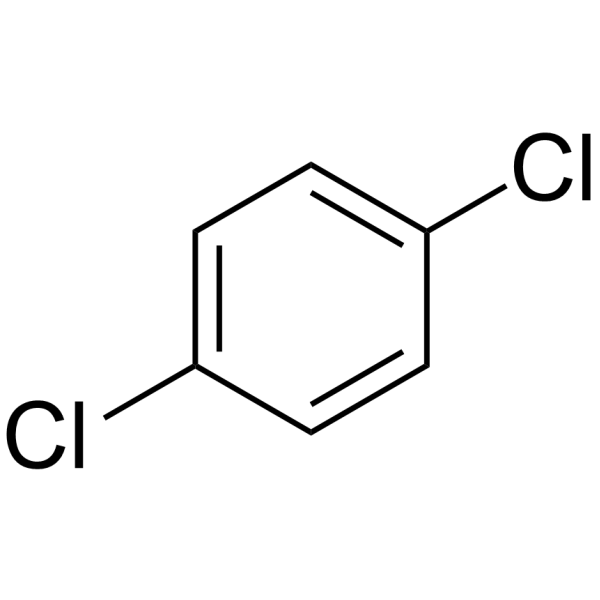
What is the name of the following compound? a) p-dichlorobenzene b) 1,4- dichlorobenzene c) phenyldichloride d) p-dichlorobenzene and 1, 4- dichlorobenzene e) 1, 4-dichlorobenzene and phenyldichlor | Homework.Study.com

Write equations for the reaction of p-dichlorobenzene with Br_2 and FeBr_3 catalyst. | Homework.Study.com

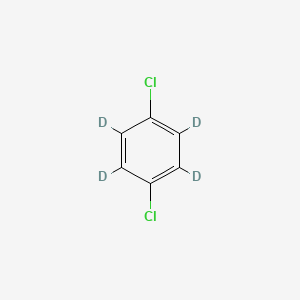
3855-82-1 | 1,4-Dichlorobenzene-d4 | Di-chloricide-d4; Dichlorocide-d4; Evola-d4; NSC 36935-d4; PDB-d4; Paradiv; Paradichlorobenzene-d4; Paradow-d4; Paramoth-d4; Persia-Perazol-d4; Santochlor-d4; p-Chlorophenyl chloride-d4; p-Dichlorobenzene-d4; para ...